|
(methyl carbanion) has tetrahedral structure (sp3) and one of the hybrid orbitals contains the lone pair of electrons. (methyl carbonium ion) also has trigonal planar structure (sp2). READ ALSO: How would you characterize Zaphod through his behavior actions?ĬH3 (methyl free radical) has a planar structure with sp2 hybridization of ‘C’ atom. As it has no lone pairs of electron pairs, the three groups to which it is bonded are arranged like a triangle around the central atom, with the bond angles measuring 120 degrees. How do you know if it is a trigonal planar?Ī compound with trigonal planar molecular geometry has a central atom bonded to three other atoms or groups. The electron nonbonding domain takes up space, making the molecule bent. Ozone has three electron groups around the central oxygen, so has the trigonal planar electron geometry, while it has a bent shape instead of a linear shape. Ozone molecule made up of three oxygen atoms, often referenced as O3. There is evidence that tiny quantities of cyclic ozone exist at the surface of magnesium oxide crystals in air. It would differ from ordinary ozone in how those three oxygen atoms are arranged. Like ordinary ozone (O3), it would have three oxygen atoms. 1b) ozone is a polar molecule (dipole moment = 0.5337 D).Ĭyclic ozone is a theoretically predicted form of ozone. READ ALSO: Why do you randomly wake up at 3am? Is ozone polar or nonpolar molecule?Īs a consequence of the bent structure, and the resonance hybridization (Figure 9.2. It is because the two oxygen atoms have same electronegativity. electrons are not displaced towards any atom. In oxygen molecule(O=O) the shared electron pairs are equally attracted by the oxygen atoms i.e. Is O3 polar or nonpolar?īecause of the molecule’s shape, the dipole moments do not cancel, so the ozone molecule is polar. Some ions with trigonal planar geometry include nitrate (NO − 3), carbonate (CO 2− 3), and guanidinium (C(NH. The structure of ozone has 3 oxygen atoms, but steric hindrance prevents it from forming a triangular structure, with each O atom forming the expected 2 bonds.Įxamples of molecules with trigonal planar geometry include boron trifluoride (BF3), formaldehyde (H2CO), phosgene (COCl2), and sulfur trioxide (SO3). 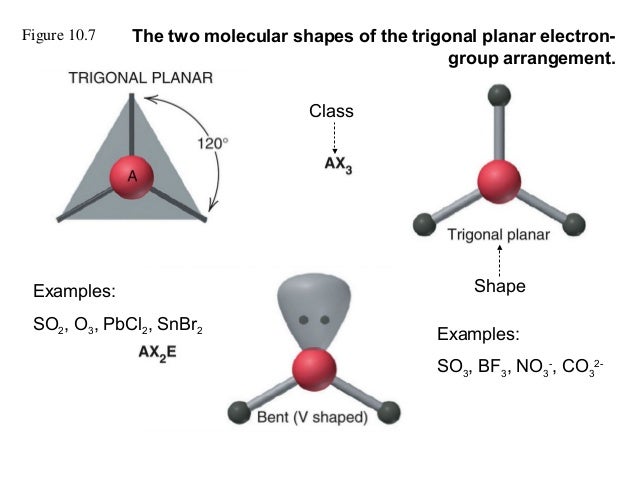
Ozone Factfile It is an allotropic form of oxygen having three atoms in each molecule, formula O3. This will result in the end O groups being pushed down giving the O3 molecule a bent molecular geometry or V shape. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
